FAQs
GENERAL QUESTIONS
How can I order Argentyn 23 Products?
Are elemental (neutral) particles of silver (AG) more effective than positively charged silver?
The scientific literature states that it is the free silver ions (i.e. the positively-charged species) that are active and that there is a "negligible particle-specific" effect. [6] In fact, researchers found that "the biological activity of nanosilver has been attributed to the associated silver cation and its soluble complexes." [7]
Why is Argentyn 23 Bio-Active Silver Hydrosol clear in color while most other silver products are not?
Silver colloids have a color based on their particle size, ranging from orange to brown to yellow to clear as their size decreases. The property is called Surface Plasmon Resonance. Particles smaller than 5nm are "subplasmonic" meaning they are clear and colorless solutions. Argentyn 23 Bio-Active Silver Hydrosol is a mixture of positively charged silver ions and silver nanoclusters with particles as small as 0.8 nm (nanometers). Color is visible when particles are longer (above 5 nm). A larger quantity of particles can also lead to visible yellow color as more particles absorb more light. Darker color is an indication of one or more of the following: even larger particles, compounds (salts or proteins), reduction of bio-active silver ions, or impurities. A smaller particle size is desirable due to greater surface area available for conversion to bio-active silver. Since visible color is an indication of larger particles, then visible color is your visual cue to less effective silver products.
Are other silver products with higher concentrations (higher ppm) more powerful?
No. More is not always better. What makes one silver product more effective than another is not its PPM concentration, but rather its particle size, positive charge, form and purity. The smaller the particle, the more bioavailable it becomes, and the greater the surface area available for conversion to positively-charged, bio-active silver while within the body.* Argentyn 23 Bio-Active Silver Hydrosol has an unprecedented particle size as small as 0.8 nanometers.
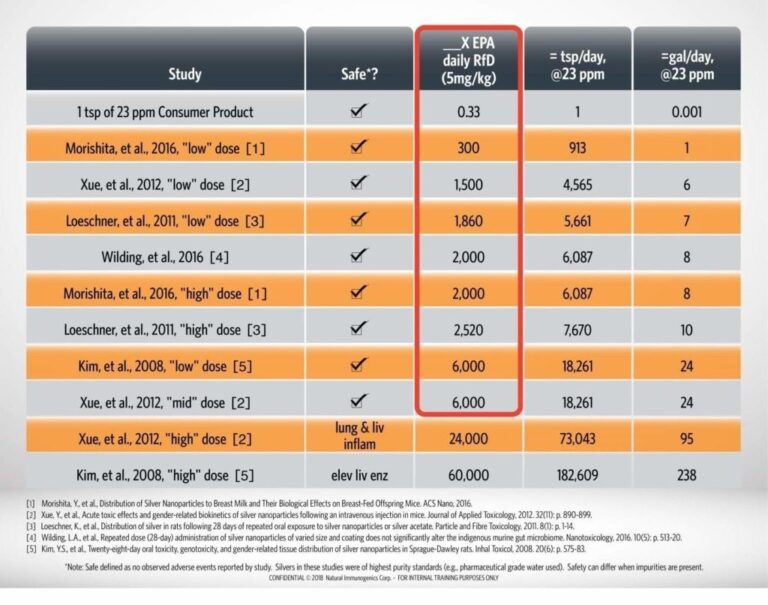
Can you provide information on safety, side effects and adverse effects of Argentyn 23 Bio-Active Silver Hydrosol?
Safety:* Argentyn 23 is marked as a dietary supplement under DSHEA. Federal agencies responsible for the protection of communities from harmful health effects related to exposure to natural and man-made hazardous substances, such as the Environmental Protection Agency (EPA) and the Agency of Toxic Substances and Drugs Registry (ATSDR) for example, have issued several risk assessments for Silver, and have established a Threshold of Toxicity Concern (TTC) of 350mcg/day for an average adult of 70Kg (for silver ingestion). Our suggested dosage of up to 3 tsp. a day as labeled, is based on these assessments; therefore, we can say that Argentyn 23 Bio-Active Silver Hydrosol is safe* when following these label directions, or use as directed by a healthcare professional. In addition to the above, a summary of peer-reviewed literature studies on ionic silver and colloidal silver (silver nanoparticles) is presented in the table below. [1-5] No observed adverse events are reported until 24,000X the EPA daily reference dose (RfD) of 5 micrograms of silver per kilogram of bodyweight. It is critical to note that all literature studies used high purity water, high purity silver materials, and endotoxin free materials. The purity of Argentyn 23 exceeds the standards used in these studies, thereby allowing interpretation of this literature with confidence. The presence of other ingredients in the formulations or other contaminants can dramatically impact the safety profile of other colloidal silver products.
SAFETY
Can long-term use of Argentyn 23 Bio-Active Silver Hydrosol damage cells?
No. In order to exhibit long term damage, mammalian cells require silver exposures that are several orders of magnitude in excess of Argentyn 23 dosages. [9][10]
Does Argentyn 23 Bio-Active Silver Hydrosol have a taste?
Argentyn 23 Bio-Active Silver Hydrosol tastes like water, although sometimes people report detecting a faint metallic taste. This faint metallic taste might feel stronger, usually due to the pH of the mouth when the product was ingested. For example, if you recently drank coffee or orange juice, which are highly acidic, you might be more apt to detect the faint metallic taste. Metabolic changes, such as acidic saliva (most likely due to changes in diet of from dietary choices) could result in a change in the pH of your saliva, which may cause Argentyn 23 to taste stronger (bitter/sour/pungent) than "water with faint metallic taste". A secondary cause could be leaching of metals from dental amalgams. However, leaching usually only occurs when the saliva is acidic, this pointing back to a dietary change. You can easily find lists of alkaline foods on the internet. Organoleptic (taste & odor) tests are part of our Quality Control batch release parameters. Our products is manufactured in an NSF-certified GMP facility in the same exact way with the same exact components, each and every time, and we have never released a "bad" batch.
Is it possible to overdose?
Not on the silver in Argentyn 23 Bio-Active silver Hydrosol. The amount of water ingested to get to a level of toxicity from silver in Argentyn 23 will cause a toxic response long before silver does. According to the peer-reviewed literature, observed acute toxicity events of liver and lung inflammation are reported at 24,000X and 60,000X the EPA Reference Dose of 5 mcg of silver per kg of body weight, which would require ingesting over 93 gallons of Argentyn 23 for a 70kg adult. [4, 5]
Can Argentyn 23 Bio-Active Silver Hydrosol cause Argyria?
No, when following label directions or as directed by a healthcare professional. Argyria can occur after a lifetime exposure to all forms of silver exceeding 10 grams for the average adult according to the World Health Organization. [8] The most dramatic symptom of argyria is the skin becoming blue or bluish-gray colored. Argyria may be found as generalized argyria or local argyria. Argyrosis is the corresponding condition related to the eye. Argentyn 23 Bio-Active Silver Hydrosol is free of proteins, salts, and other compounds and therefore poses no health hazard when used as directed.* However other colloidal silvers may contain these impurities therefore increasing the risk of causing argyria. Products greatly exceed the EPA oral Daily Reference Dose (RfD) for silver of 350 mcg (micrograms). Argentyn 23 Bio-Active Silver Hydrosol is the only professional silver supplement to have received a statement of safety from Dr. Dana Flavin; Founder and Executive Director; former Science Assistant to the Associate Bureau Director, Division of Toxicology, US FDA, Washington DC.
EFFICACY
Can you provide me information on clinical usefulness?
Many reports of immune support benefits have been received from satisfied patients and practitioners alike. Under the Dietary Supplement Health and Education Act (DSHEA), we cannot make specific marketing claims about diagnosing, treating, mitigating or curing any disease or condition.
GUT MICROBIOME
What does Argentyn 23 Bio-Active Silver Hydrosol do in the gut? How does it affect good and bad bacteria?
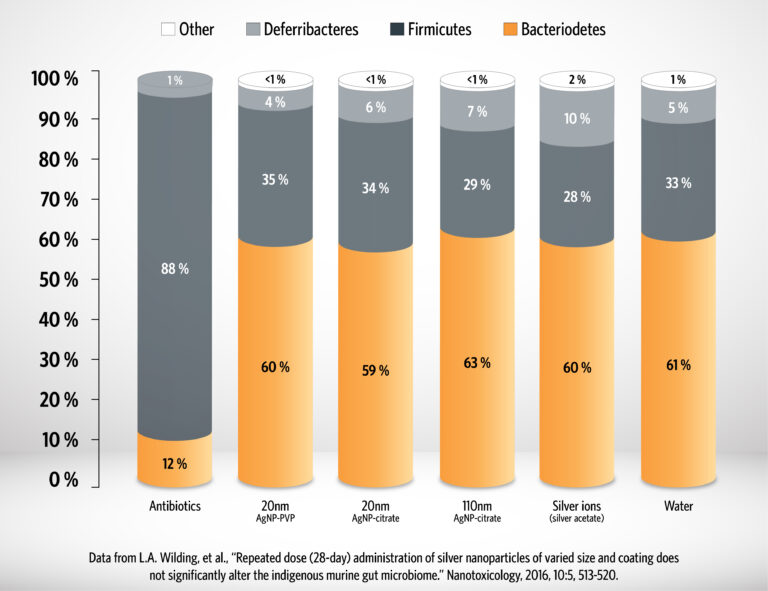
In accordance with the terms of the Wilding et al. study and as discussed above, we have no reason to believe that Argentyn 23 would alter the gut microbiome diversity.*[4] According to Wilding et al., both good and bad bacteria are impacted equally, whereas prescription antibiotic cefoperazone disrupts the balance of bacterial species dramatically. [4] Therefore, it is sometimes recommended to supplement with beneficial probiotics in the opposite time of day as Argentyn 23 is taken, assist with the balance of good bacteria in the gut microbiome.
Figure data from [4], Wilding, et al., (2016). "Repeated dose (28-day) administration of silver nanoparticles of varied size and coating does not significantly alter the indigenous murine gut microbiome." Nanotoxicology 10(5): 513-520.
Figure data from [4], Wilding, et al., (2016). "Repeated dose (28-day) administration of silver nanoparticles of varied size and coating does not significantly alter the indigenous murine gut microbiome." Nanotoxicology 10(5): 513-520.
Sovereign Copper Professional
What is copper, and why is it important?
Copper is a mineral that you need to stay healthy. Your body uses copper to carry out many important functions, including making energy, connective tissues synthesis, and blood vessels. Copper also helps maintain the nervous and immune systems and activates genes. Your body also needs copper for brain development. For more information, visit the Copper Fact Sheet from the National Institute of Health found here https://ods.od.nih.gov/factsheets/Copper-HealthProfessional/#h2 (accessed 10/24/2024).
What are the health benefits of taking Sovereign Copper Professional?
Copper is a critical compound of enzyme activity in the body. As a result, many functions depend on it to:
1. Support strong and flexible arteries cartilage and connective tissue
2. Slow Build collagen for skin elasticity. Promoting healthy skin, hair and nails
3. Support healthy cardiovascular function
4. Improve energy and metabolism
5. Absorb iron and aid in red blood cell production
Furthermore, copper is recognized by Canada as a trace mineral necessary for the maintenance of good health, to help form red blood cells and to help produce and repair connective tissue.
*These statements have not been evaluated by the FDA. This product is not intended to diagnose, treat, cure, or prevent any disease.
What foods are a good source of copper?
Copper is naturally found as a trace element in foods such as organ meats, oysters, shellfish, whole grains, leafy greens, nuts, dark chocolate. People on restrictive diets, such as gluten-free, dairy-free, or nut-free may not get the typical trace quantities found in the diet.
Who should consider taking copper supplements?
Until recently, it was generally believed that most people consumed adequate quantities of copper. However, modern research has shown that this is not the case. In the United Kingdom and the United States for example, many typical meals have been analyzed for their metals content. According to recent surveys, only 25% of the US population consumes the amount of copper a day estimated to be adequate by the US Food and Nutrition Board of the National Academy of Sciences. Typical diets in the US provide only about half of this amount and some diets in mainly industrialized countries contain less than 40% of the recommended dietary allowance. In the United Kingdom, it is now recommended that the daily intake should range from 0.4mg/day for 1-3-year-old children to 1.2mg/day for adults – see here for more related information: https://www.copper.org/consumers/health/cu_health_uk.html
How much copper should I take daily?
Maintenance:
Adults: One tablespoon daily (450mcg)
Children 4 years and older: 2 teaspoons (300mcg)
Long Term Support:
Adults: 2 tablespoons daily (900mcg)
Short Term support (Copper "Quick Fix”):
Adults: 3 tablespoons daily (1350mcg)
Natural Immunogenics markets its Copper Hydrosol product as a Dietary Supplement and therefore, is to be used only for children 4 years and above. The form of Copper in the product is approved by federal risk assessment agencies for the claims the product carry. The maintenance dosage information is safe at 51% of the RDI per dose for adults, or 68% of the daily value for children 4 years and over.
What are the ingredients in Sovereign Copper?
Just 3 ingredients: 99.999% pure copper, pharmaceutical grade water (USP-NF) and a hint of pure silver.
What are the ingredients in Sovereign Copper?
Sovereign Copper has a concentration of 10ppm ideal for maintaining healthy copper levels, while Sovereign Copper professional has a concentration of 30ppm ideal for people diagnosed with copper deficiency, or who need a “quick copper fix”.
Does Sovereign Copper Professional interact with medications or other dietary supplements?
Copper is not known to interact with any medications. However, it’s always important to tell your doctor, pharmacist, and other health care providers about any dietary supplements and prescription or over-the-counter medicines you take. They can tell you if the dietary supplements might interact with your medicines or if the medicines might interfere with how your body absorbs, uses, or breaks down nutrients such as copper.
Does the product have a flavor?
No, Sovereign Copper Professional is flavorless to a slight metallic taste.
How should the product be stored?
For best shelf-life, avoid strong sources of electromagnetic energy and direct sunlight. Product can also be kept refrigerated after opening if necessary but in general, can remain stable at standard room temperature.
Is this product Gluten free or vegan?
Yes, Sovereign Copper Professional is gluten free, allergen free, vegan and non-GMO.
What form do copper supplements come in?
This product is available for sale in 2oz, 4oz and 8oz and will soon become available in 16 and 32 bottle sizes.
What makes Sovereign Copper Professional different than other copper supplements?
Sovereign Copper Professional Bio-Active Hydrosol is a proprietary blend of positively-charged (bio-active) Copper (I) & Copper (II) species. There are parts of the body that preferentially utilize each of the forms of copper to support various vital functions. Sovereign Copper's Dual Action Copper (I) & Copper (II) proprietary blend facilitates effective enzymatic activity necessary to support important vital body functions. Sovereign Copper is more bio-available to the body eliminating the extra work that must be done to convert it from one state to the other.
What is the source of your copper ingredient?
The source is Elemental Copper of a purity level of ≥99.999% (ultra-pure). Copper species are generated from the unique and proprietary manufacturing process using pharmaceutical grade purified water meeting USP standards.
Can I dilute Sovereign Copper Professional? Can I mix it with saline or juices?
Dilution is not recommended, since the product contains a safe, low concentration of copper. We strongly recommend against mixing it with saline or other potential binding agents.
Is Sovereign Copper Professional safe for pregnant or nursing women?
Although copper is an essential mineral required of the human and animal diet and the RDI for pregnant or nursing women is in fact higher than the 900mcg/day however, FDA regulations limit the labeling of dietary supplements. Please consult with your health care provider to find out if copper hydrosol is appropriate for your needs during pregnancy or for the needs of your baby. Copper is naturally found as a trace element in foods such as organ meats, oysters, shellfish, whole grains, leafy greens, nuts, dark chocolate.
Can you use Sovereign Copper Professional in cats and dogs?
Yes. In fact, a certain amount of copper is required in dog food. According to the official 2015 AAFCO publication, dog food must contain no less than 7.3 mg of copper per kilogram of dog food.
AAFCO also recommends 1-1.5 mg of copper daily for cats.
Can I take Sovereign Copper Professional and Sovereign Silver together?
Sovereign Copper has been specifically formulated so that, even if taken together with Sovereign Silver, the Silver dosage remains below or within the margin of safety defined by the EPA for orally ingested silver
Are there any contra-indications to taking this product?
Yes, in case of Wilson’s disease. People with this condition cannot consume copper because their bodies are unable to properly process and excrete copper. This genetic disorder leads to an accumulation of copper in the liver, brain, and other tissues, which can cause serious health problems, including liver damage, neurological issues, and psychiatric symptoms. Limiting copper intake is crucial to prevent further accumulation and manage the disease effectively. Avoiding copper-rich foods and supplements is a key part of the treatment plan for individuals with Wilson's disease. Our product labels mention this.
Previous: Publications
Next: Regulatory Compliance
REFERENCES
- Morishita, Y., et al., Distribution of Silver Nanoparticles to Breast Milk and Their Biological Effects on Breast-Fed Offspring Mice. ACS Nano, 2016.
- Xue, Y., et al., Acute toxic effects and gender-related biokinetics of silver nanoparticles following an intravenous injection in mice. Journal of Applied Toxicology, 2012. 32(11): p. 890-899.
- Loeschner, K., et al., Distribution of silver in rats following 28 days of repeated oral exposure to silver nanoparticles or silver acetate. Particle and Fibre Toxicology, 2011. 8(1): p. 1-14.
- Wilding, L.A., et al., Repeated dose (28-day) administration of silver nanoparticles of varied size and coating does not significantly alter the indigenous murine gut microbiome. Nanotoxicology, 2016. 10(5): p. 513-20.
- Kim, Y.S., et al., Twenty-eight-day oral toxicity, genotoxicity, and gender-related tissue distribution of silver nanoparticles in Sprague-Dawley rats. Inhal Toxicol, 2008. 20(6): p. 575-83.
- Xiu, Z.-m., et al., Negligible Particle-Specific Antibacterial Activity of Silver Nanoparticles. Nano Letters, 2012. 12(8): p. 4271-4275.
Liu, J., et al., Controlled Release of Biologically Active Silver from Nanosilver Surfaces. ACS Nano, 2010. 4(11): p. 6903-6913. - Organization, W.H., Health criteria and other supporting information. Guidelines for drinking-water quality, 2nd ed. Vol. 2. 1996, Geneva.
- Lee, J.H., et al., Biopersistence of silver nanoparticles in tissues from Sprague–Dawley rats. Particle and Fibre Toxicology, 2013. 10(1): p. 36.
- Zumwalde, R., E. Kuempel, and G. Holdsworth, External Review Draft – Current Intelligence Bulletin: Health Effects of Occupational Exposure to Silver Nanomaterials., C.f.D.C.a.P. U.S. Department of Health and Human Services, National Institute for Occupational Safety and Health, Editor. 2015, NIOSH: Cincinnati, OH.
